- #Graphpad prism 4 parameter logistic curve serial
- #Graphpad prism 4 parameter logistic curve software
Enter Y values with no X values in those rows (example below), or X values with no Y values in those rows.
Enter dataĮnter the unknowns below the standards on the same table. In this case, if you want to take into account variations in the SD from point to point, use nonlinear regression to fit the line. You can also choose to enter data where the mean and SD (or SEM) have already been calculated. Choose replicate values if you have replicates to enter. If you are entering your own data, choose the subcolumn format. If you are just getting started, choose the sample data: Interpolate unknowns from a linear standard curve 4PL assumes symmetry around the inflection point while 5PL takes asymmetry into account, which normally is a better fit for immunoassays.From the Welcome or New Table dialog, choose to create an XY data table.
#Graphpad prism 4 parameter logistic curve software
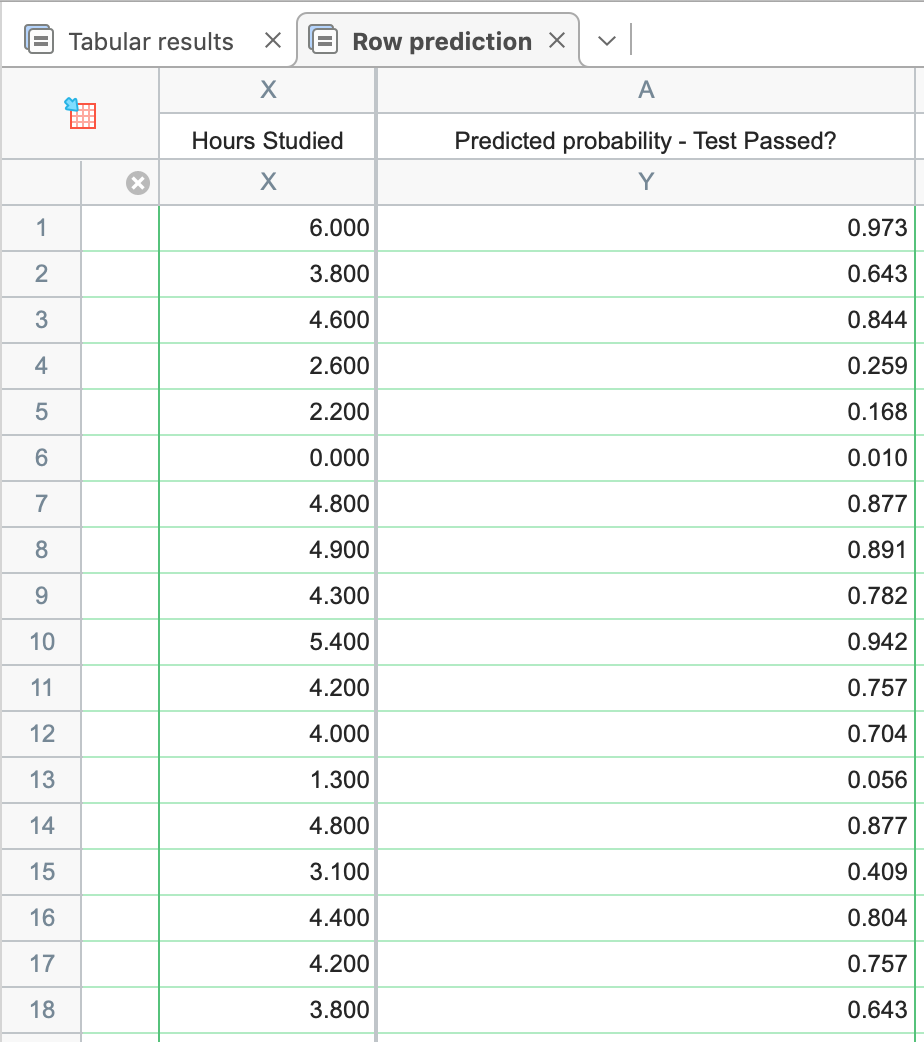
The concentration of the antigen in the sample is calculated by extrapolation of the linear portion of the standard curve.įigure 1: Example of a quantitative ELISA standard curve from Human ICAM1 SimpleStep ELISA® Kit (ab174445).Ĭurve fitting software allow using different models to plot your data. Most ELISA plate readers will incorporate a software for curve fitting and data analysis. The standard curve is derived from plotting known concentrations of a reference antigen against the readout obtained for each concentration (usually optical density at 450 nm). The standard or calibration curve is the element of the quantitative ELISA that will allow calculating the concentration of antigen in the sample. This is the most commonly reported ELISA data.
#Graphpad prism 4 parameter logistic curve serial
It requires comparison of the values measured for the samples with a standard curve prepared from a serial dilution of a purified antigen in a known concentration. Quantitative ELISA allows calculating the amount of antigen present in the sample.
Semi-quantitative ELISA allows the relative comparison of the antigen levels between the samples.It requires a blank well containing no antigen or an unrelated control antigen.

